Reversible Na Plating/Stripping with High Areal Capacity Using an Electroconductive Liquid Electrolyte System
- Journal
- ACS Applied Materials & Interfaces
- Year
- 2023
- Link
- https://pubs.acs.org/doi/full/10.1021/acsami.3c06554 914회 연결
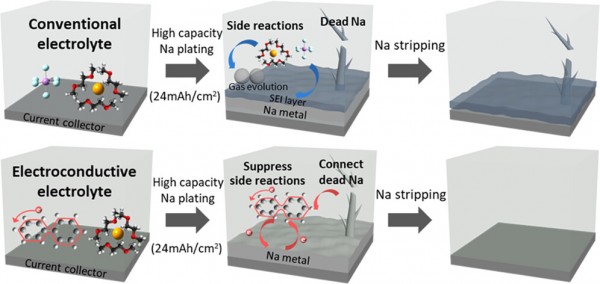
Anode-free sodium-metal batteries (AFSMBs) are promising candidates for maximizing energy density and minimizing cost and safety hazards in the absence of metallic sodium during cell assembly. The practical implementation of AFSMBs is hindered by the low cycling stability of Na-metal plating and stripping, particularly under high areal capacities, due to unstable solid electrolyte interphase (SEI) layer formation with electrolyte decomposition and inactive dead Na formation. Here, we proposed an electroconductive electrolyte system consisting of liquid electrolytes that accept electrons at a certain energy level and form electronically conductive and solid electrolytes that prevent internal short circuit through low electronic conductivity. The electron acceptability and high electronic conductivity of the liquid electrolyte can suppress the irreversible electron transfer with electrolyte decomposition and reutilize the inactive dead metal, respectively. The functions of the system were demonstrated using a sodium biphenyl liquid electrolyte–NASICON solid electrolyte in a seawater battery (SWB) system, which features an infinite sodium source. The anode-free SWB cells achieved a high Coulombic efficiency of ≥99.9% for over 60 cycles at a high areal capacity of ∼24 mAh/cm2. This study provides insight into the Na plating/stripping properties in anode-free systems and proposes a significant strategy for improving the reversibility of metal anodes for various battery systems with solid electrolytes.