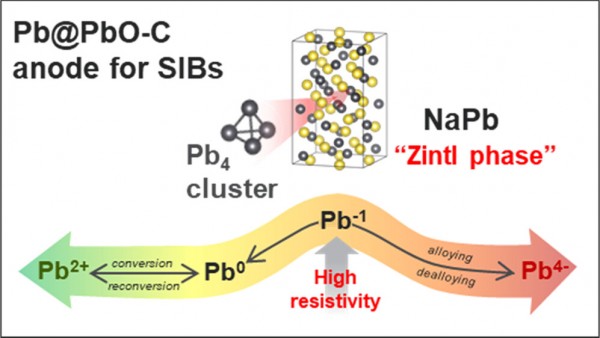
Evidence of Zintl Intermediate Phase and Its Impacts on Li and Na Storage Performance of Pb-Based Alloying Anodes
- Journal
- Chemistry of Materials
- Year
- 2023
- Link
- https://doi.org/10.1021/acs.chemmater.3c00061 931회 연결
Anode materials based on conversion and alloying reactions are promising to achieve high energy density of advanced sodium-ion batteries (SIBs). While the chemical similarities between sodium and lithium as alkali elements make the benchmarking strategy practical in developing new high-performance anodes, simply borrowing the anode material from one system to the other does not always guarantee success unless it is based on sound understanding of both Li- and Na-reaction mechanisms. In this work, we report the Na storage performance of a Pb-based anode and its fundamental reaction dynamics. In contrast to its excellent electrochemical performances in Li cells (reversible ∼600 mAh/g), the newly developed Pb@PbO–C nanocomposite anode has limited electrochemical Na reaction properties showing moderate capacity and rate performances (∼300 mAh/g at 20 mA/g). Synchrotron-based X-ray diffraction and absorption spectroscopy studies reveal the fundamental differences in the Na and Li reaction mechanism of the Pb-based anode. Unlike Li reaction, the unique Na reaction mechanism involves the formation of a highly ionic NaPb Zintl phase, which comprises tetrahedral Pb4 clusters, as an intermediate phase. The strong covalent character of the Pb4 Zintl clusters adversely affects the electronic conductivity and thus limits the electrochemical performance of the Pb-based anode in Na cells. These findings provide new insights applicable to developing high-performance alloying anode materials.