Unveiling interfacial dynamics and structural degradation of solid electrolytes in a seawater battery system
- Journal
- Journal of Materials Chemistry A
- Vol
- 8
- Page
- 21804-21811
- Year
- 2020
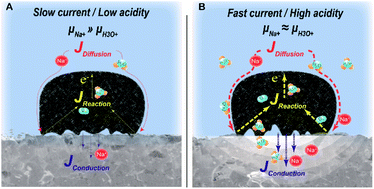
Understanding the interfacial reaction between a solid electrolyte and reactive species is of vital importance for the development of various battery systems. In particular, the interaction between the solid electrolyte, aqueous media, and dissolved gaseous phases at catalysts affects reaction kinetics and hybrid battery performance. These effects are concentrated at the interfaces that the three kinetic fluxes simultaneously encounter, which we refer to as a “triple-flux interface (TFI)”. The TFI consists of a Na+ super ionic conductor as a solid electrolyte, a seawater catholyte, and dissolved gaseous phases on a catalytic carbon felt. Here, we provide insights into the interfacial dynamics during the operation of a hybrid Na-seawater battery under harsh operating conditions, including at high current densities and in acidified seawater solutions. We discover that high current densities and the permeation of acid compounds during the charge process cause the irreversible extraction of Na+, crevasse-like brokenness, and unavoidable phase distortion from the surface that is in contact with seawater. The investigation of the factors in terms of chemical potentials of the charge carriers would provide insight into designs for high-powered seawater battery systems.