Tetraruthenium Polyoxometalate as an Atom-Efficient Bifunctional Oxygen Evolution Reaction/Oxygen Reduction Reaction Catalyst and Its Application in Seawater Batteries
- Journal
- ACS Applied Materials & Interfaces
- Vol
- 12
- Page
- 32689-32697
- Year
- 2020
- Link
- https://pubs.acs.org/doi/full/10.1021/acsami.0c07225 1162회 연결
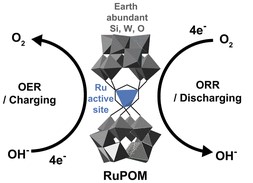
Although development and utilization of efficient catalysts with earth-abundant and cheap elements are desired, precious noble metal-based catalysts are still widely used and studied due to the urgent need to address energy and environmental issues. Polyoxometalates (POMs) can be excellent candidates in this context. In this study, we found that oxo-bridged tetraruthenium polyoxometalate (RuPOM) exhibits excellent electrocatalytic activity for both oxygen evolution and reduction reactions (OER and ORR) with minimal use of noble metal elements and can be used for the development of efficient seawater batteries (SWBs). The deposition of RuPOM on a desired electrode with conducting carbon Ketjen black (KB) by the simple slurry coating method imparted bifunctional OER/ORR activity to the underlying electrode. Although the mass activity was similar, RuPOM/KB mixtures exhibited superior activity even compared to commercially available Pt/C when comparing the activity per noble metal element. Based on these findings, we employed RuPOM to develop efficient SWBs. RuPOM significantly lowered the charging potential and increased the discharging potential of SWBs, which are related to OER and ORR, respectively. This study can provide insights into the development of POM-based electrocatalysts and their application in energy storage and conversion devices.