Structural characterization of layered Na0.5Co0.5Mn0.5O2 material as a promising cathode for sodium-ion batteries
- Journal
- Journal of Power Sources
- Vol
- 363
- Page
- 442-449
- Year
- 2017
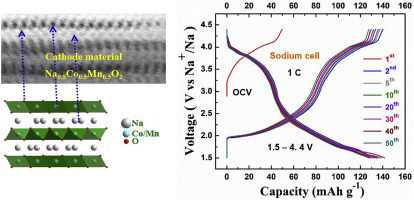
Layered Na0.5Co0.5Mn0.5O2 material is synthesized through a facile mixed hydroxy-carbonate route using (Co0.5Mn0.5)2(OH)2CO3 precursor and well characterized as a hexagonal layered structure under P63/mmc space group. The lattice parameters and unit cell volume (a = 2.8363 Å, c = 11.3152 Å and V = 78.83 Å3) are calculated by Rietveld refinement analysis. A flaky-bundle morphology is obtained to the layered Na0.5Co0.5Mn0.5O2 material with the hexagonal flake size ~ 30 nm. Advanced transmission electron microscopic images are revealed the local structure of the layered Na0.5Co0.5Mn0.5O2 material with contrasting bright dots and faint dark dots corresponding to the Co/Mn and Na atoms. Two oxidation and reduction peaks are occurred in a cyclic voltammetric analysis corresponding to Co3+/Co4+ and Mn3+/Mn4+ redox processes. These reversible processes are attributed to the intercalation/de-intercalation of Na+ ions into the host structure of layered Na0.5Co0.5Mn0.5O2 material. Accordingly, the sodium cell is delivered the initial charge-discharge capacity 53/144 mAh g–1 at 0.5 C, which cycling studies are extended to rate capability test at 1 C, 3 C and 5C. Eventually, the Na-ion full-cell is yielded cathode charge-discharge capacity 55/52 mAh g–1 at 0.212 mA and exhibited as a high voltage cathode for Na-ion batteries.